Mycobacterium leprae, the cause of leprosy
Issue: Mycobacteria
27 August 2014 article
Leprosy is a chronic infectious disease caused by Mycobacterium leprae. It damages peripheral nerves and can affect the skin, eyes, nose and muscles. Nerve injury in leprosy can cause severe disabling deformities.
Although it is often thought of as a disease of ancient times, it still occurs today and the World Health Organization recorded 3.8 million new cases of leprosy from 105 different countries in the last decade. Leprosy is only rarely reported in Europe today, but it was once prevalent throughout the region and may still occur among people who live or work abroad in endemic countries. People with leprosy often suffer profound social stigma on account of their disease, and leprosy imposes tremendous economic and psychological burdens on individuals and healthcare systems.
Leprosy (also known as Hansen’s Disease) is considered a Neglected Tropical Disease. It is a rare infection, usually found in fewer than 1:10,000 people in most populations, and often associated with poverty. The largest numbers of new cases today originate in South-east Asia, the Americas and Africa. Leprosy is curable with multiple antibiotic therapy, usually consisting of rifampin, dapsone and clofazimine. Early detection and treatment can help avoid many of the disabling complications of leprosy. Although several prototype assays are in development, there are no current laboratory screening tests to aid early detection of leprosy, and the disease must be diagnosed clinically.
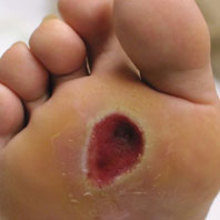
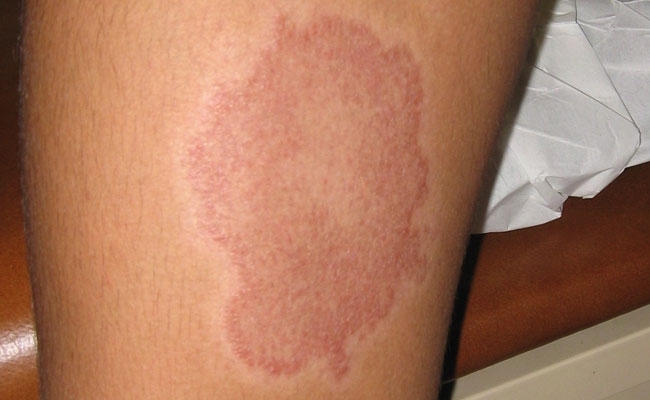
FIG. 1. BORDERLINE TUBERCULOID LEPROSY LESION ON LOWER EXTREMITY. THE CENTRE OF THIS LESION WAS ANAESTHETIC.
Clinical features
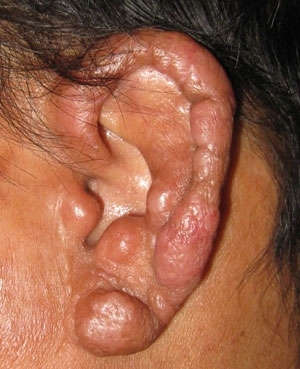
Leprosy is not highly contagious. Susceptibility to leprosy appears to be genetically based and 95% of all people appear to be naturally resistant to the disease. People who develop leprosy usually incubate the infection for 3–5 years before manifesting illness, and they exhibit a broad spectrum of clinical and histopathological responses to the infection determined by their immunological response to Mycobacterium leprae. In the tuberculoid portion of the spectrum, people have few discrete lesions consisting of well-organised granulomas with small numbers of bacilli (Fig. 1). While those in the lepromatous portion of the spectrum may have several nodular or diffuse lesions (Fig. 2) containing many bacilli in poorly organised granulomas. However, the most important pathological feature common to all forms of leprosy is involvement and damage of the peripheral nervous system (PNS).
FIG. 2. LEPROMATOUS LEPROSY LESION MANIFEST ON EAR. ABUNDANT INTRACELLULAR ACID-FAST BACILLI WERE PRESENT IN THIS TISSUE, A COOL SITE FAVOURABLE FOR GROWTH OF M. LEPRAE.
Nerve injury
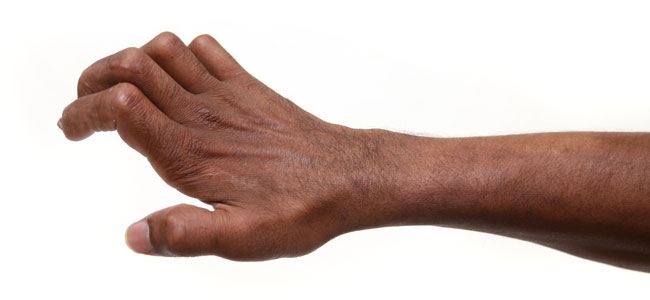
M. leprae has the remarkable capacity to invade Schwann cells of the peripheral nerve, which enclose and support the axons of sensory and motor neurons. Schwann cell infection causes many complex biological and pathological alterations including demyelination, de-differentiation and reprogramming of the cells. Axons unprotected by Schwann cells are vulnerable to injury and may be destroyed by the host’s inflammatory response to M. leprae. Generally, small unmyelinated and myelinated sensory neurons are affected first, and as the disease progresses motor neurons and muscles may also be compromised. Patients lose sensation in areas affected by leprosy. They often cannot discern the difference between hot and cold, and anaesthesia in the hands and feet contribute significantly to pressure ulceration (Fig. 3, top) and traumatic injury and secondary infections, which may lead to the loss of digits and other serious consequences. If not interrupted by treatment, nerve injury may progress to motor weakness and paralysis of intrinsic muscles in fingers or toes, causing life-long disability (Fig. 4).
FIG. 4. CLAWED HAND RESULTING FROM ADVANCED INJURY TO THE ULNAR NERVE DUE TO LEPROSY.
There are still large gaps in our understanding of neuropathogenesis of leprosy. Peripheral nerves not only serve as the principal target for M. leprae infection, but also serve as a safe haven for the bacillus since the blood–nerve barrier protects the organism from many host immune responses. M. leprae appears to take advantage of the remarkable regenerating capacity of the adult PNS in securing its preferred niche, and regeneration of damaged peripheral nerves even continues after treatment in patients with advanced leprosy. Nerve injury may progress gradually over the entire course of the disease and may be irreversible.
Intracellular parasite
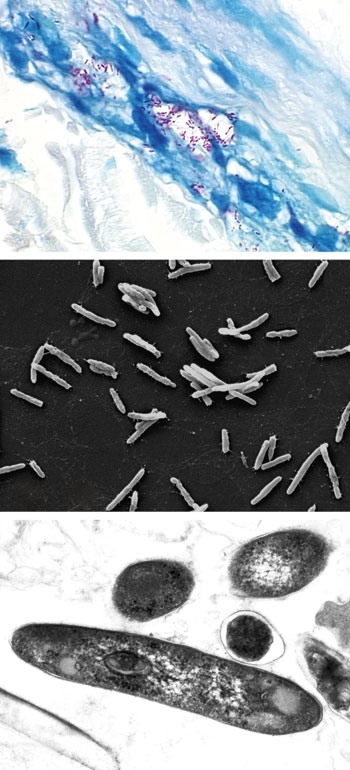
M. leprae is a highly adapted niche pathogen. It is weakly acid-fast (Fig. 5). Its genome has undergone drastic reductive evolution. At only 3.3Mbp it has the smallest genome among the mycobacteria, and non-functional pseudogenes also occupy nearly half the chromosome. This loss of functionality probably underlies our continuing inability to culture the organism on artificial laboratory media, and probably influences the organism’s remarkably long generation time (12.5 days). M. leprae prefers cool temperatures. Lesions on patients are usually found in cooler areas of the skin. Viability of M. leprae decreases quickly at temperatures above 35°C. Most animals readily clear the bacilli and cannot be experimentally infected with M. leprae. Limited replication can be achieved after inoculation of M. leprae into the hind foot pads of conventional and athymic mice. However, the bacilli will not grow when inoculated systemically and exponential increases in growth require many months to attain. Infection in mice does not involve the animal’s nerves. The only animal that reliably recapitulates leprosy as seen in humans, and develops extensive neurological involvement with M. leprae, is the nine-banded armadillo (Dasypus novemcinctus). Other than humans, armadillos are also the only natural hosts of M. leprae.
FIG. 5. (A) FITE-STAINED SECTION OF SKIN SHOWING M. LEPRAE (RED) IN A CUTANEOUS NERVE, THE PATHOGNOMONIC FEATURE OF LEPROSY PATHOLOGY. (B) SCANNING ELECTRON MICROGRAPH OF M. LEPRAE. BAR, 5 ΜM. (C) TRANSMISSION ELECTRON MICROGRAPH OF M. LEPRAE IN CROSS SECTION. BAR, 250 NM.
Armadillos
Armadillos are exotic-looking animals about the size of house-cats (Fig. 6). With thick, tough skin and a hard, flexible carapace armouring most of their body, Rudyard Kipling suggested that armadillos were a blend of a tortoise and a porcupine. They are mammals of the order Xenarthra, and related to sloths and anteaters. Their normal body temperature ranges from 33 to 35°C, and it was this trait that first attracted the attention of leprosy researchers. Experimental infection of armadillos with M. leprae requires 18–24 months to manifest as a fully disseminated disease, but prolific quantities (1012) of bacilli can be harvested from a single animal, and armadillos are the hosts-of-choice for propagation of leprosy bacilli. The remarkable quantities of M. leprae made available through armadillos have been a boon to leprosy research.
Shortly after initial discovery of the armadillo’s unique susceptibility to experimental infection, a naturally occurring, systemic mycobacteriosis was also found among free-ranging armadillos in the USA. Subsequent surveys confirmed that wild armadillos are a large reservoir for M. leprae and the animals had harboured a natural infection with M. leprae for many decades prior to their ever being used in leprosy research. Leprosy was not present in the New World during pre-Colombian times, and it is reasonable to assume that armadillos must have acquired the infection from humans sometime in the last few centuries. They are now recognised as the only non-human reservoir of M. leprae, and are part of the natural ecology of the disease in the USA. Recent reports indicate that zoonotic transmission of M. leprae from armadillos is responsible for up to 64% of all leprosy cases among persons born in the USA. Armadillos range from the southern USA through Latin America to Argentina, and biomarkers for M. leprae have been observed among some South American armadillos. The role that armadillos may play in perpetuating leprosy in the Americas is now being investigated.
FIG. 6. NINE-BANDED ARMADILLLO (DASYPUS NOVEMCINCTUS).

Vaccine and diagnostics
M. leprae is usually thought to be transmitted from person-to-person by respiratory routes, and current global control strategies focus on antibiotic treatment of active cases to cure the infection and interrupt transmission. However, evidence is accumulating that an occult reservoir of pre-clinical asymptomatic cases, and perhaps armadillos or other environmental hosts, may play a more important role in the ecology of the disease, and leprosy probably cannot be eliminated through drug therapy alone. Control of leprosy by vaccination or immunotherapy as an adjunct to drug therapy may have significant advantages over control by drug treatment alone. A consortium of philanthropic foundations have promoted research on new diagnostic tests and a prototype vaccine since 2002. Scientists from all over the world are engaged in these efforts and their products are now beginning to move into trial. If technology triumphs, we may finally put an end to the suffering caused by one of the oldest known mycobacterial diseases.
RICHARD TRUMAN, RAHUL SHARMA, MARIA PENA, BARBARA STRYJEWSKA, JOHN FIGAROLA & DAVID SCOLLARD
United States Department of Health and Human Services, Health Resources and Services Administration, Healthcare Systems Bureau, National Hansen’s Disease Program, Baton Rouge, Louisiana, USA
[email protected]
FURTHER READING
Masaki, T. & others (2013). Reprogramming adult Schwann cells to stem cell-like cells by leprosy bacilli promotes dissemination of infection. Cell 152, 51–67.
Monot, M. & others (2009). Comparative genomic and phylogeographic analysis of Mycobacterium leprae. Nat Genet 41, 1282–1289.
Rodrigues, L. C. & Lockwood, D. (2011). Leprosy now: epidemiology, progress, challenges, and research gaps. Lancet Infect Dis 11, 464–470.
Scollard, D. M. & others (2006). The continuing challenges of leprosy. Clin Microbiol Rev 19, 338–381.
Truman, R. W. & others (2011). Probable zoonotic leprosy in the southern United States. N Engl J Med 364, 1626–1633.
Image: Fig. 1. Barbara Stryjewska. Fig. 2. Barbara Stryjewska. Fig. 3. Plantar pressure ulceration due to anaesthesia is a common sequela of nerve injury in the lower extremity in leprosy. Dane Hupp. Fig. 4. Alicia Hoard. Fig. 5. Greg McCormick. Fig. 6. Greg McCormick..
