Farm management and antimicrobial resistance in Northern Ireland
Posted on August 4, 2021 by Katie Lawther
In this blog, Katie, a third year PhD student in the Huws Lab at Queen’s University Belfast discusses her research into antimicrobial resistance in Northern Ireland and how this is influenced by farm management practices. Katie presented her research at the Microbiology Society’s Annual Conference Online 2021.
Part of the Institute for Global Food Security, our lab is mainly focused on understanding the functionality of the microbes in the forestomach – or rumen – of cattle, with the aim of addressing food security and human health. A part of this involves antimicrobial resistance (AMR) surveillance and monitoring, which is where I fit in.
As part of my microbiology degree, I completed my undergraduate research project in the Huws lab researching novel antimicrobial peptides; this is when Professor Sharon Huws and I began to discuss future PhDs. During this time and throughout my undergraduate degree I developed a real interest in the agri-food side of microbiology. With almost as many cows as people in Northern Ireland, I was very fortunately surrounded by many beautiful farmlands when growing up there. As a nation, we take great pride in our farming and produce. As I progressed through my degree, I first discovered the ‘One Health’ approach and the idea of different research sectors working together in the interest of better public health. Through this, I learnt that I could combine my different areas of interest including human health and agri-food microbiology.
As part of my PhD, our research mainly looks at AMR in Northern Irish agriculture. Agriculture forms a huge part of life on the isle of Ireland and within the UK, but AMR threatens food security, as well as both animal and human health. To assess this risk, we are performing surveillance work on AMR within Northern Irish dairy farms. This includes the prevalence of resistant micro-organisms within dairy calves and the environment they are exposed to. We aim to not only determine and describe these AMR levels, but also to identify calf exposures and identify farming practices that can lead to increased or decreased AMR levels. By identifying such practices, we hope to create better guidelines for the agriculture sector on how to limit their on-farm resistance.

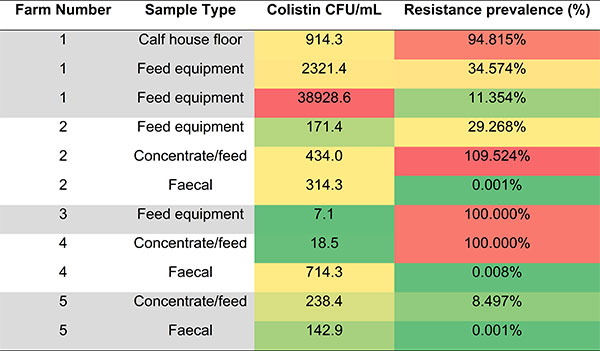
To achieve this, we collected samples from dairy farms across the country. These were then processed using a combination of wet-lab culture-based techniques focusing on phenotypic resistance and bioinformatic approaches looking at the complete resistome. Culture-based techniques focused on resistance against the following antibiotics: amoxicillin, erythromycin, florfenicol, neomycin, tetracycline and trimethoprim. These antimicrobials were chosen because they are commonly used in agriculture. Colistin resistance was also investigated due to its importance in human healthcare. Resistance prevalence was determined in the lab and the resistant isolates will soon undergo identification through sequencing. In contrast, the samples also underwent shotgun metagenomic sequencing. This work is ongoing but aims to identify the resistome (including the resistant unculturable organisms) as well as understand the context of AMR genes including their association with mobile genetic elements.

Experiments are still ongoing, however early findings have shown that the dairy farm environment does contain high levels of resistance and a wide variety of resistance was seen between and within each farm. Colistin resistance was detected in half of the farms sampled. On farms with colistin resistance, food and feed equipment could potentially be acting as AMR reservoirs.
This research can highlight how to improve farm management practices and how they impact AMR. By working with the NI government, we hope to increase and strengthen our AMR research. The future of this work would be to form a better understanding of the resistome including the resistance mechanisms and assess the risk of future threats including emerging zoonotic resistant disease and horizontal gene transfer. We hope by completing this research we can not only determine the AMR levels but offer solutions to the problem by assisting farmers to reduce AMR and antibiotic usage while maintaining the high levels of animal welfare that they pride themselves on. The research has been very diverse which is exciting; the mixture of wet-lab techniques and bioinformatics is both challenging and enjoyable. The fact this work could one day impact real farmers and people is very rewarding. AMR is a complex, ever-changing narrative but this means that the research is extremely interesting, and it is amazing to see the incredible research going on in this field.
Thank you to my supervisors and all of the Huws lab that have been involved in this project. Thanks also to my collaborators from the Optihouse project and my funders, DAERA NI.
For any questions about this research project, contact Katie Lawther at [email protected].

