Implementation of Next-Generation Sequencing-Based HIV Drug Resistance Testing: Enhancing Antiretroviral Treatment Monitoring and Public Health Outcomes
Human Immunodeficiency Virus (HIV) continues to pose a significant global health challenge despite advances in antiretroviral therapy (ART). The emergence of drug-resistant HIV strains remains a critical obstacle in the effective management of HIV infections. Traditional methods of detecting HIV drug resistance (HIVDR) have limitations in sensitivity and scalability. Next-generation sequencing (NGS) technology offers a promising solution, providing detailed insights into the viral genome and enabling the detection of drug resistance mutations at much lower frequencies than conventional methods. Public health laboratories are exploring the implementation of NGS-based HIVDR testing.
The need for enhanced HIVDR testing
ART has transformed HIV from a fatal disease to a manageable chronic condition. However, the long-term success of ART is threatened by the development of drug resistance. The World Health Organisation (WHO) report in 2024 indicated increased resistance to dolutegravir or DTG (an HIV integrase inhibitor drug and one of the most prescribed ART for treating people living with HIV infection). The WHO offered recommendations for countries to develop and implement effective approaches of monitoring and responding to HIVDR in infected populations. HIV's high mutation rate allows it to rapidly evolve, potentially rendering standard treatments ineffective. Traditional HIVDR testing methods, such as Sanger sequencing, have been instrumental in identifying resistance mutations but are limited in their ability to detect low-frequency variants that can emerge under selective ART drug pressure in HIV-infected individuals.
NGS offers a more sensitive and comprehensive approach to HIVDR testing. Unlike Sanger sequencing, which can miss minority drug resistant variants present at less than 20% of the viral population in HIV-infected individuals, NGS can detect viral variants at frequencies as low as 1%. This capability is crucial for early detection of drug resistance mutations, which can inform timely adjustments to treatment regimens, thereby enhancing the effectiveness of ART and reducing the likelihood of treatment failure.
Implementation of NGS-based HIVDR testing
The implementation of NGS-based HIVDR testing involves several key steps like sample preparation, sequencing, data analysis, and clinical interpretation.
Sample preparation begins with the extraction of viral Ribonucleic acid (RNA) from patient samples, followed by reverse transcription to generate complementary Deoxyribonucleic acid (cDNA). The cDNA is then amplified using polymerase chain reaction (PCR) to target specific regions of the HIV genome known to harbour drug resistance mutations.
Sequencing involves fragmenting the cDNA and attaching indexes and adapters necessary for subsequent processes. NGS platforms, such as Illumina, Pacific Biosciences (PacBio), Thermo Fisher Scientific, Oxford Nanopore Technologies and BGI Genomics, are then used to sequence the prepared DNA. These platforms generate vast amounts of complex genomic data, covering the viral genome at high depth.
Data analysis comprises use of bioinformatics tools to assemble the viral genome and identify mutations. Specialised software is employed to distinguish true resistance mutations from sequencing errors and to quantify the frequency of each mutation within HIV viral population.
Clinical interpretation ensures that the detected mutations are interpreted in the context of their known impact on drug resistance. This involves consulting databases and guidelines, such as those provided by the Stanford HIV Drug Resistance Database, to determine the clinical significance of each mutation and recommend appropriate changes to the patient's HIV treatment regimen.
Benefits for NGS-based HIVDR monitoring
NGS-based HIVDR testing offers several benefits for the monitoring and management of ART such as.
- Early detection of drug resistance: The high sensitivity of NGS allows for the early detection of low-frequency drug resistance mutations, which can emerge before clinical failure is evident. This early warning system enables healthcare providers to adjust ART regimens proactively, preventing the expansion of resistant strains.
- Comprehensive ART resistance profiles: NGS provides a complete picture of the viral population, identifying both major and minor resistance mutations. This comprehensive profile is essential for tailoring ART strategies to the specific resistance patterns present in each patient.
- Enhanced surveillance: NGS can be used for routine surveillance of HIVDR at the population level, identifying trends in resistance mutations and informing public health strategies. This information is vital for updating national treatment guidelines and optimizing the allocation of resources for HIV care.
Why we developed HIV-DRIVES: HIV Drug Resistance Identification, Variant Evaluation, and Surveillance bioinformatic pipeline
Despite its advantages, the implementation of NGS-based HIVDR testing faces several challenges. The cost and infrastructure to deploy NGS platforms and the associated bioinformatics infrastructure are expensive and require significant investment. The technical expertise: the complexity of NGS genomic data generated necessitates specialised technical expertise, which may be lacking in resource-limited settings though available analysis platforms either require payment for subscription, may not be easily interoperable or may not be secure for transmission of patient data as national laws may necessitate. The use of NGS involves the generation and handling of vast amounts of genetic data, raising concerns about patient privacy and data security. Ethical considerations such as removing patient’s genetic data before analysing the viral data must be addressed to ensure the responsible use of NGS technology. Beyond capacity building and training, the interpretation of NGS data requires robust databases and standardised guidelines. Continuous updates to resistance databases and the development of automated interpretation tools can help streamline this process, and to address these challenges, we developed and implemented HIV-DRIVES at the National Genomics Reference Laboratory.
Public health implications
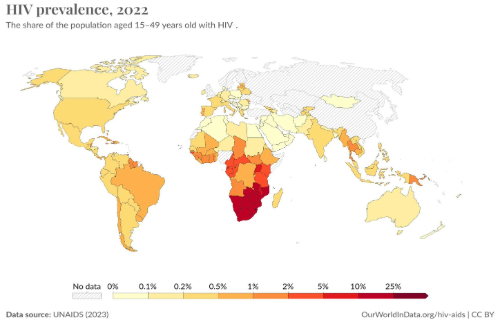
The implementation of NGS-based HIVDR testing has significant implications for public health. It enables personalised treatment adjustments based on detailed resistance profiles. Furthermore, NGS enhances the effectiveness of ART, leading to better health outcomes for individuals living with HIV. In the global fight to eradicate HIV epidemic, early detection and management of drug resistance can reduce the transmission of resistant HIV strains. This is particularly important in high-prevalence settings where the spread of resistant viruses can compromise the efficacy of first-line ART regimens. It is also important to noted that data generated from NGS-based HIVDR testing can inform public health policies and resource allocation. For instance, identifying regions with high levels of specific resistance mutations can guide the distribution of alternative ART drugs. COVID-19 pandemic hurled NGS platforms to many public health laboratories in low- and middle-income countries (LMICs) providing an opportunity to rapidly scaleup and enhance HIVDR monitoring and improve treatment outcomes in these settings. We are convinced that implementing NGS-based HIVDR testing in LMICs (where more than 80% of all people living with HIV lived in 2020) can significantly impact global HIV control efforts.
Way forward
In our study, we introduce HIV-DRIVES - an easy-to-use NGS-HIVDR bioinformatics pipeline for ART drug resistance profiling as well as determining HIV subtypes. The implementation of NGS-based HIV drug resistance profiling represents a significant advancement in the management of HIV infection. By providing detailed and sensitive detection of drug resistance mutations, NGS enhances ART monitoring and improves patient outcomes. Moreover, the comprehensive resistance profiles generated by NGS have important implications for public health, informing treatment guidelines, and reducing the transmission of resistant strains. Despite the challenges associated with its implementation, the potential benefits of NGS-based HIVDR testing make it a crucial tool in the ongoing global fight to end HIV/AIDS epidemic.
Image: Generated by Gerald Mboowa using the ImagineArt text-to-image AI model
