Double Trouble: When Two Top-Priority Pathogens Meet
I am Orlando Ross, a PhD candidate at the Medical Research Council’s Centre for Medical Mycology, based at the University of Exeter. I am a researcher of parasite and fungal immunology, with a focus on co-infections. This interest in co-infections peaked during my MRes Medical Mycology and Fungal Immunology (the first year of my current 1+3 MRC CMM MRes-PhD programme). One of my projects was to investigate Cryptococcus neoformans-Mycobacterium tuberculosis co-infections, supervised by Dr. Liliane Mukaremera and Dr. Ivy Dambuza. The work undertaken during this time formed the basis of our newly published paper, “Cryptococcus neoformans responds to presence of mycobacterium by diversifying its morphologies and remodelling its capsular material”.
As a group, we developed a novel method for culturing Cryptococcus neoformans in a human plasma-like media (HPLM) which is more physiologically representative than traditional media (such as YPD, which has an abundance of nutrients for the fungus to grow). We have positioned our research to mimic host environments, so that our findings could be as translational as possible to host biology.
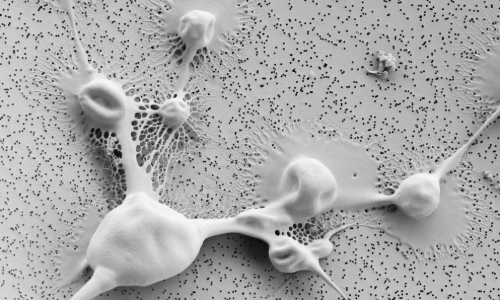
A scanning electron microscope image of Cryptococcus neoformans surface to highlight how viscous and thick the outer capsule is, and how capsule masks the cell wall from detection by immune cells. Credit: Liliane Mukaremera (MRC CMM), Christian Hacker and Lenka Koptasikova (University of Exeter Bioimaging)
Cryptococcus neoformans is a fungal pathogen responsible for nearly 200,000 deaths every year. C. neoformans is responsible for nearly 20% of all HIV-related deaths, and is a ubiquitous opportunistic pathogen, found in pigeon guano across the world, and eucalyptus trees. The WHO has classified C. neoformans as a critical priority fungal pathogen, with an HIV-associated mortality of nearly 70%.
Mycobacterium tuberculosis (MTb) is a bacterium that, until the SARS-CoV-2 pandemic was the leading single-agent cause of death worldwide. MTb has a mortality of 50% in untreated patients and is thought to be responsible for 1.7 billion latent infections globally, with prevalence in people living with HIV-AIDS.
Both pathogens form granulomas in the lung, which can then rupture resulting in dissemination – in the case of C. neoformans this often locates to the central nervous system whereby cryptococcal meningitis results in devastating disease and presentation. This stage is usually where symptoms occur and is often too late for treatment.
As both pathogens establish infection in the lung, and have similar epidemiological overlap, we began to wonder if there would be any interaction between the two. We know that both C. neoformans and MTb are recognised by similar receptors in the body, but could these pathogens also influence each other? Could these pathogens alter host response to each other? That’s what we set out to understand.
Early on in this project, we cultured the C. neoformans reference strain with heat-killed MTb (as MTb is such a dangerous pathogen, we are not cleared to work with a live-strain in our lab). We noticed that C. neoformans cells appeared bigger and with larger capsule and were really excited by this observation – I was looking down the microscope at the slides and had to go and find Ivy to show her. We did a little dance to celebrate this finding and then decided to test other isolates (both high and low virulence clinical isolates) and live M. bovis (the vaccination strain that is used for BCG vaccines) to test multiple avenues and see if these observations were antigen-specific or cell wall mediated.

Orlando and Ivy celebrating their first observations of C. neoformans capsule being significantly larger when co-cultured with M. tuberculosis. Credit: Orlando Ross.
Amazingly, we saw that isolates responsible for increased mortality produced significantly higher numbers of yeast and titan cells, whereas those C. neoformans isolates associated with host survival produced significantly more small cells; a morphology linked to improved dissemination and no titan cells. Together, this shows us that C. neoformans isolates alter their morphology along a trajectory that correlates with virulence.
We also observed that 75% of clinical isolates produced significantly larger capsule when grown in the presence of BCG. Capsule is a key component of this fungi’s immune evasion and persistence in the host. This capsule remodelling was isolate-specific, instead of correlating with virulence, indicating to us that the capsule remodelling program is a plastic process with key implications for co-infection with tuberculosis-causing pathogens.
Finally, we wanted to translate these findings to implications on host immunology. As such, we used a novel protocol to generate alveolar-like macrophages (AMs). AMs have key differences to a traditional in vitro macrophage and closely mimic true alveolar macrophages. This setting fits within our approach to model host biology, compared to traditional cell lines (such as THP-1) or generating macrophages from bone marrow (BMDMs).
Once we had these AMs developed, we spiked them with our heat-killed MTb and the cytokine IFN-γ. We did this to replicate the lung microenvironment of an active tuberculosis infection. We showed that this tuberculosis-like environment actually increased the numbers of internalised C. neoformans cells but that these cells were not killed. This suggested that C. neoformans can persist and proliferate within AMs during tuberculosis infection and take advantage of this environment.
This was really exciting because it showed for the first time, the implications of tuberculosis-causing pathogens on morphology and immune consequences of C. neoformans. Aside from sparse case-reports of co-infections between these species, we believe we are the first people in the world to show that these pathogens interact. Whilst our focus was on C. neoformans’ perspective, we are excited by this research and hope that this will pave the way for a new field of investigations.
We hope, long-term, that this research could encourage enhanced screening in patients with tuberculosis infections for other pathogens, especially C. neoformans. Early detection and treatment of cryptococcosis directly correlates with improved survival, and we envisage that this field of research can improve patient outcomes and survival, lessen the burden of disease and raise awareness of the importance in considering fungal infections.
Whilst I have moved on to research helminth infections and the implications of these on host control of fungal colonisation and infection, Liliane continues her excellent work on C. neoformans capsule, and Ivy has launched a career of research into C. neoformans-M. tuberculosis co-infections. I am really proud of the role we played in kickstarting this avenue of exciting and cutting-edge research.